What are Congenital Disorders of Glycosylation?
Congenital Disorders of Glycosylation (CDG) are inheritable metabolic disorders that effect gylcosylation. There are 42 different enzymes that have been implicated in CDG, but the most common form of CDG, PMM2-CDG or CDG-1a, is caused by a mutation in the gene PMM2. Over 70% of known cases of CDG are due to missense mutations in PMM2 [1].
How are patients with CDG affected?
What is Glycosylation?
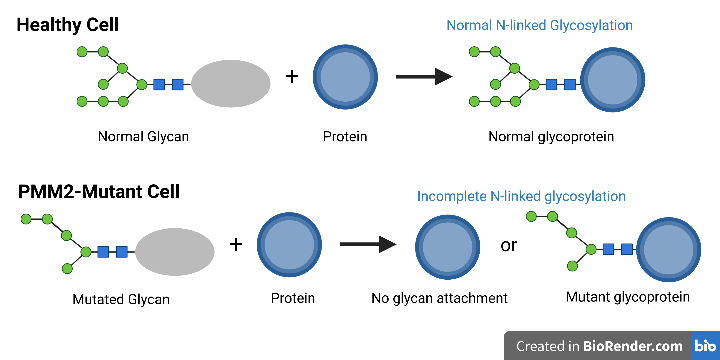
Glycosylation is the the process cells use to build the long sugar chains that are attached to glycoproteins [2]. When glycosylation genes are mutated, it can cause no sugar chain to be attached to the protein, or for a mutated form to be attached. The addition of sugar chains, called glycans, to the proteins affect the final 3D structure of a protein [3]. The shape of a protein affects the ability of a protein to function within the body [3].
|
What Does PMM2 Do?
|
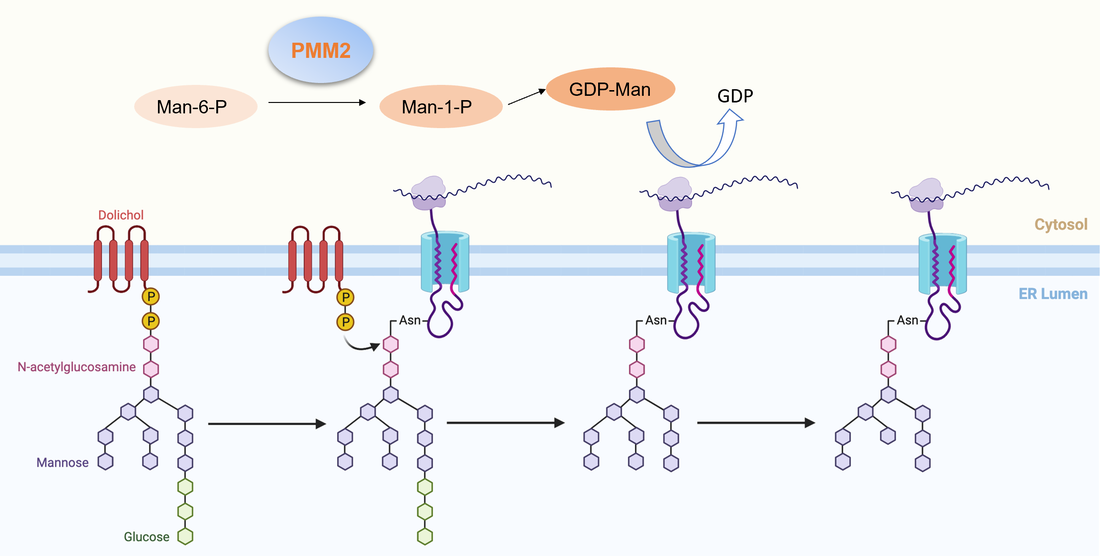
PMM2, or phosphomannomutase 2, is a gene found on chromosome 16p13 [6]. Missense mutations in this gene cause defects in the sugar building block pathways of the endoplasmic reticulum [6]. PMM2 converts mannose-6-phosphate to mannose-1-phosphate, which creates GDP-mannose [5]. These are precursors to the oligosacccaride chain, or glycan [5]. These defects result in a mutated glycan, which can either cause attachment failure to proteins or incomplete glycosylation [3]. When N-linked glycans are unable to attach, or are incomplete, they do not perform the same functions [3]. This pathway also provides energy to the cell.
|
What does a PMM2 mutation affect?
|
PMM2 mutations cause defects in many bodily functions, as N-linked glycosylation is essential for life [4]. In most animal models, complete knockout of PMM2 results in lethality [7,8]. Researchers have had to adapt their models in order to have viable test subjects [8,9]. Patients diagnosed with PMM2-CDG can have varying clinical presentations, based on which mutations they have in the gene [6].
|
What are the gaps in knowledge?
- How does PMM2 function in cardiac development?
- Can supplementing patients with mannose-1-phosphate allow their N-linked glycosylation pathways to function normally? Could a therapy involving this rescue the disease phenotype?
- Why do most cases of CDG have neuronal issues?
- How do different therapeutic approaches work with different mutations in the PMM2 gene?
Where Can I Learn More?
|
|
|
References
1. National Institute of Health. Congenital Disorders of Glycoslyation. Rare Diseases. [Online] National Institute of Health, 2020. https://rarediseases.info.nih.gov/diseases/10307/congenital-disorders-of-glycosylation.1.
2. Congenital Disorders of Glycosylation. Jaeken, Jaak and Matthijs, Gert. s.l. : Annual Review of Genomics and Human Genetics , 2001, Vol. 2. https://doi.org/10.1146/annurev.genom.2.1.129
3. Brian J. Arey (September 26th 2012). The Role of Glycosylation in Receptor Signaling, Glycosylation, Stefana Petrescu, IntechOpen, DOI: 10.5772/50262. Available from: https://www.intechopen.com/books/glycosylation/the-role-of-glycosylation-in-receptor-signaling.
4. CDG Care. What is CDG? CDG Care. [Online] 2020. http://cdgcare.com/what-is-cdg/.
5. Markus Aebi,N-linked protein glycosylation in the ER,Biochimica et Biophysica Acta (BBA) - Molecular Cell Research,Volume 1833, Issue 11,2013, Pages 2430-2437,ISSN 0167-4889, https://doi.org/10.1016/j.bbamcr.2013.04.001.
6.Congenital Disorders of N-Linked Glycosylation and Multiple Pathway Overview. Sparks, Susan E and Krasnewich, Donna M. s.l. : GeneReviews, January 12, 2017.
7. Thiel C, Lübke T, Matthijs G, von Figura K, Körner C. Targeted disruption of the mouse phosphomannomutase 2 gene causes early embryonic lethality. Mol Cell Biol. 2006 Aug;26(15):5615-20. doi: 10.1128/MCB.02391-05. PMID: 16847317; PMCID: PMC1592760.
8. Parkinson WM, Dookwah M, Dear ML, Gatto CL, Aoki K, Tiemeyer M, Broadie K. Synaptic roles for phosphomannomutase type 2 in a new Drosophila congenital disorder of glycosylation disease model. Dis Model Mech. 2016 May 1;9(5):513-27. doi: 10.1242/dmm.022939. Epub 2016 Mar 3. PMID: 26940433; PMCID: PMC4892659.
9. Jessica P Lao, Nina DiPrimio, Madeleine Prangley, Feba S Sam, Joshua D Mast, Ethan O Perlstein, Yeast Models of Phosphomannomutase 2 Deficiency, a Congenital Disorder of Glycosylation, G3 Genes|Genomes|Genetics, Volume 9, Issue 2, 1 February 2019, Pages 413–423, https://doi.org/10.1534/g3.118.200934
Header Image: Adapted from Abeomics, drawn by Taylor Mateyka